|
7/8/2023 0 Comments Atomic structure powerpoints 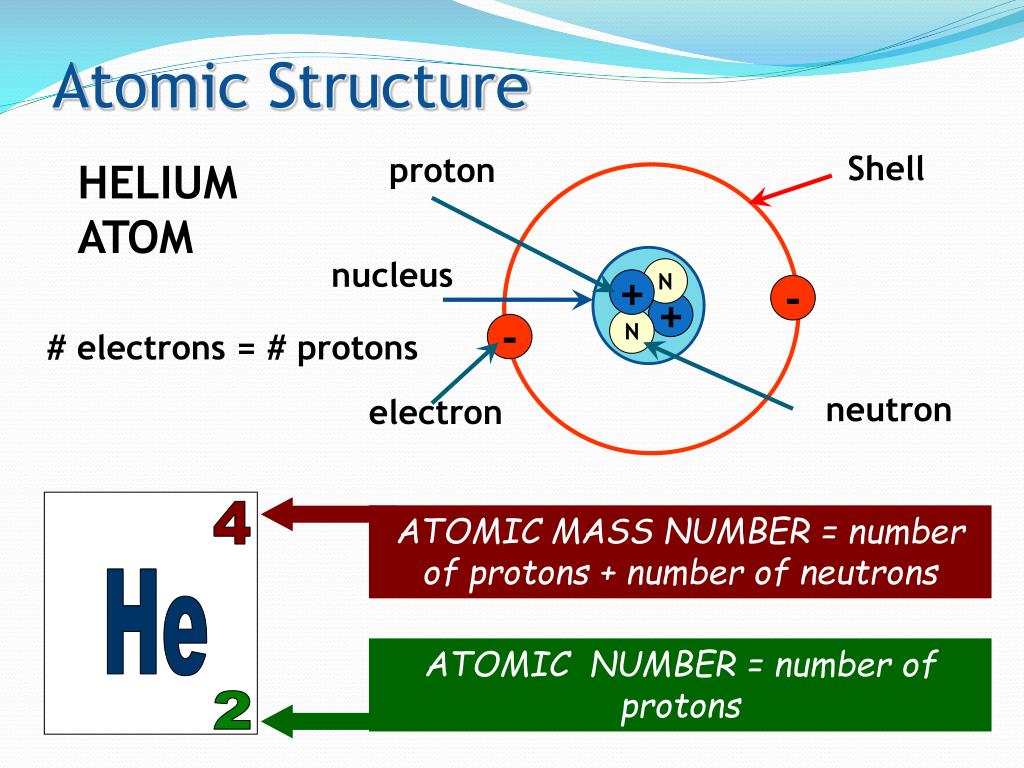
For a perspective about their relative sizes, consider this: If the nucleus were the size of a blueberry, the atom would be about the size of a football stadium ( Figure 2.11).įigure 2.11 If an atom could be expanded to the size of a football stadium, the nucleus would be the size of a single blueberry. The diameter of an atom is on the order of 10 −10 m, whereas the diameter of the nucleus is roughly 10 −15 m-about 100,000 times smaller. The nucleus contains the majority of an atom’s mass because protons and neutrons are much heavier than electrons, whereas electrons occupy almost all of an atom’s volume. It was learned that an atom contains a very small nucleus composed of positively charged protons and uncharged neutrons, surrounded by a much larger volume of space containing negatively charged electrons. The development of modern atomic theory revealed much about the inner structure of atoms. Calculate average atomic mass and isotopic abundance.Define the atomic mass unit and average atomic mass.Write and interpret symbols that depict the atomic number, mass number, and charge of an atom or ion.Our product offerings include millions of PowerPoint templates, diagrams, animated 3D characters and more.By the end of this section, you will be able to: is brought to you by CrystalGraphics, the award-winning developer and market-leading publisher of rich-media enhancement products for presentations. Then you can share it with your target audience as well as ’s millions of monthly visitors. We’ll convert it to an HTML5 slideshow that includes all the media types you’ve already added: audio, video, music, pictures, animations and transition effects. You might even have a presentation you’d like to share with others. 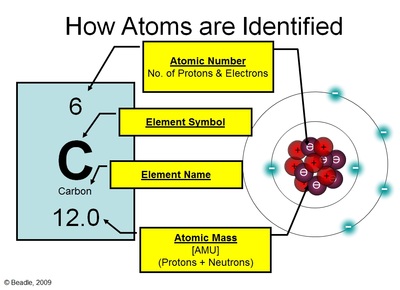
And, best of all, it is completely free and easy to use. Whatever your area of interest, here you’ll be able to find and view presentations you’ll love and possibly download. It has millions of presentations already uploaded and available with 1,000s more being uploaded by its users every day. is a leading presentation sharing website. You Practice Atomic Structure Worksheet Part Bįollow Aufbau to write electron configurations 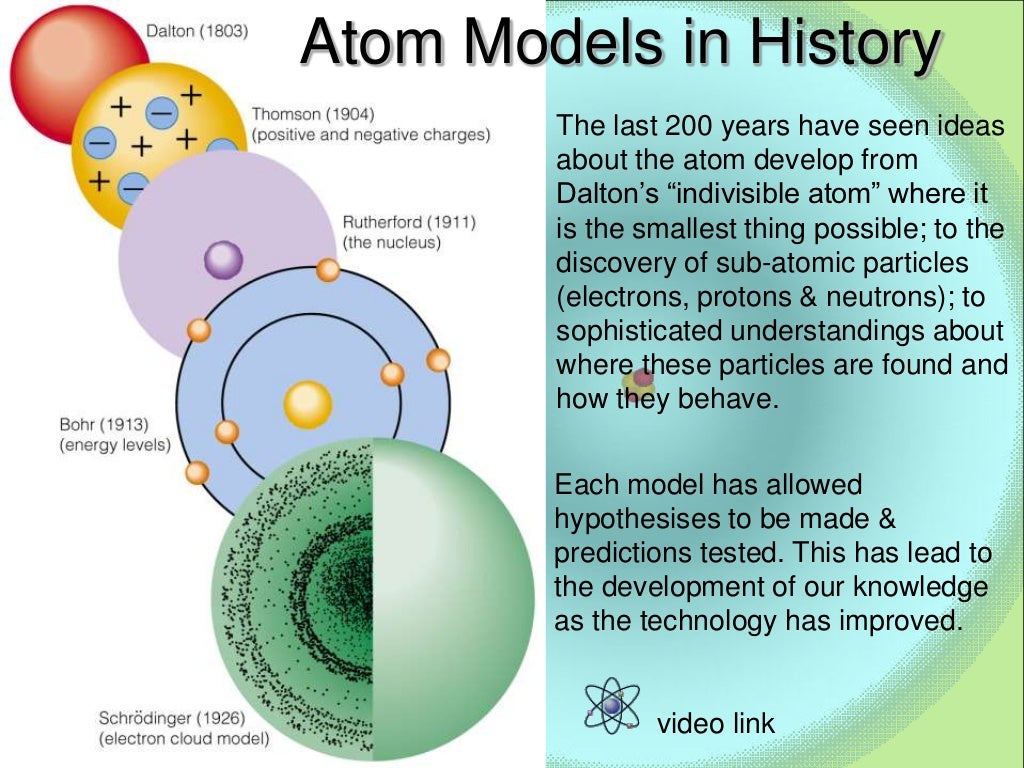
Isotopes atoms of same element with a different.Quantum mechanics mathematically describes.e- energy limited to specific values (Bohr).Cannot know both location and velocity of e.Heisenberg stated Uncertainty Principle.Still use his suggested energy values, not.Neils Bohr suggests e- orbit nucleus as planets.How studying light helps us understand atoms 5 Each color in bls has a specific ? and E given.Unique bls unique set of ? unique energies.Each element has unique bright line spectrum.How studying light helps us understand atoms 4 E of photon found by using Plancks eqn E h ?.e- in metal absorb light as particles not as.How studying light helps us understand atoms 3 electrons leave the metal as electricity.When light of specific ? shines on metal.How studying light helps us understand atoms 2 Said energy flows in packets called quanta. 
How studying light helps us understand atoms 1 Has specific colors because it has specific ?.Produced when white light goes through prismĬontinuous spectrum vs.Particle Location Charge Relative Mass(amu) Actual MassĮlectron (e-) Orbitals around nucleus 1- 0 9.11 x 10 -28 g The work of many scientists reveals a still.atom has small nucleus 1/10,000 size ofĪnyone ever accuse you of having an empty head?.Most a - Particles went straight through ?.Positive a-Particles were deflected slightly?.Cambridge Physics - Discovery of the ElectronĪ-particles at gold (1911)Rutherford's gold.Light rays glow from cathode (-) to anode ()Ĭathode Rays being deflected by magnetic field.4-Atoms combine in whole number ratios.ĭaltons Atomic Theory.3-Atoms cannot be created or destroyed.What would happen if you chopped up matter into. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
